 
Modeling the oligomeric arrangement of subunit c in the F1FO ATP synthase from the structure of the monomeric subunit and cross-linking in the native-enzyme. The impact of direct refinement against proton chemical shifts on protein structure determination by NMR. The impact of direct refinement against 13Cα and 13Cβ chemical shifts on protein structure determination by NMR. Temperature dependence of 1H chemical shifts in proteins. Crystallography & NMR system: A new software suite for macromolecular structure determination. Torsion-angle molecular dynamics as a new efficient tool for NMR structure calculation. 1H, 13C, and 15N assignments and secondary structure of the high pH form of subunit c of the F1FO ATP synthase. Proton-translocating carboxyl of subunit c of F1FOH +-ATP synthase: the unique environment suggested by the p Ka determined by 1H NMR. Heteronuclear three-dimensional NMR spectroscopy of the inflammatory protein C5a. ATP synthase: an electrochemical transducer with rotatory mechanics. Heteronuclear 1H- 15N nuclear magnetic resonance studies of the c subunit of the Escherichia coli F1FO ATP synthase: assignment and secondary structure. NMR studies of subunit c of the ATP synthase from Propiogenium modestum in dodecylsulfate micelles. Matthey,U., Kaim,G., Braun,D., Wüthrich,K. Solution structure of the transmembrane H +-transporting subunit c of the F1FO ATP synthase. Subunit interactions coupling H + transport and ATP synthesis in F1FO ATP synthase. Defining the domain of binding of F1 subunit ε with the polar loop of FO subunit c in the Escherichia coli ATP synthase. The γ subunit in the Escherichia coli ATP synthase complex (ECF1FO) extends through the stalk and contacts the c subunits of the FO part. The subunit δ-subunit b domain of the Escherichia coli F1FO ATPase. Topology, organization, and function of the psi subunit in the FO sector of the H +-ATPase of Escherichia coli. Cross-linking and labeling of the Escherichia coli F1FO-ATP synthase reveal a compact hydrophilic portion of FO close to an F1 catalytic subunit. Interacting helical faces of subunits a and c in the F1FO ATP synthase of Escherichia coli defined by disulfide cross-linking. Arrangement of the multicopy H +-translocating subunit c in the membrane sector of the Escherichia coli F1FO ATP synthase. 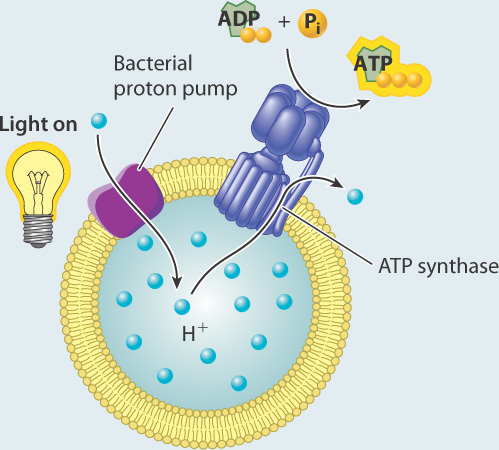
Topographical structure of membrane-bound Escherichia coli F1FO ATP synthase in aqueous buffer. Molecular imaging of Escherichia coli F1FO-ATPase in reconstituted membranes using atomic force microscopy. The FO complex of the Escherichia coli ATP synthase: Investigation by electron spectroscopic imaging and immunoelectron microscopy. Genetic fusions of subunit c in the FO sector of H +-transporting ATP synthase. Direct observation of the rotation of F1-ATPase. Intersubunit rotation in active F-ATPase. Rotation of subunits during catalysis by Escherichia coli F1 ATPase. The ATP synthase-a splendid molecular machine. The 2.8-Å structure of rat liver F1-ATPase: Configuration of a critical intermediate in ATP synthesis/hydrolysis. Structure at 2.8 Å resolution of F1-ATPase from bovine heart mitochondria. 
Where $n = 8$ for the mammalian ATP synthase.Abrahams,J. \newcommand) \simeq 12$ kcal/mol, which is about 20 times the thermal energy ($RT$ or $k_BT$) and quite a significant amount of energy!įor ATP synthesis to proceed, we know the total $\dgtot$ in (2) must be negative, so the change in proton chemical potential must be large and negative: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
